Discover the difference
The DBS battery (also called an IPG) is like a pacemaker for the brain. It sends electrical signals to specific areas of the brain to help control symptoms. There are two different types of DBS batteries: rechargeable and non-rechargeable.
Both options provide the same therapeutic benefit; the main factors for consideration are device size and the effort required to maintain therapy.
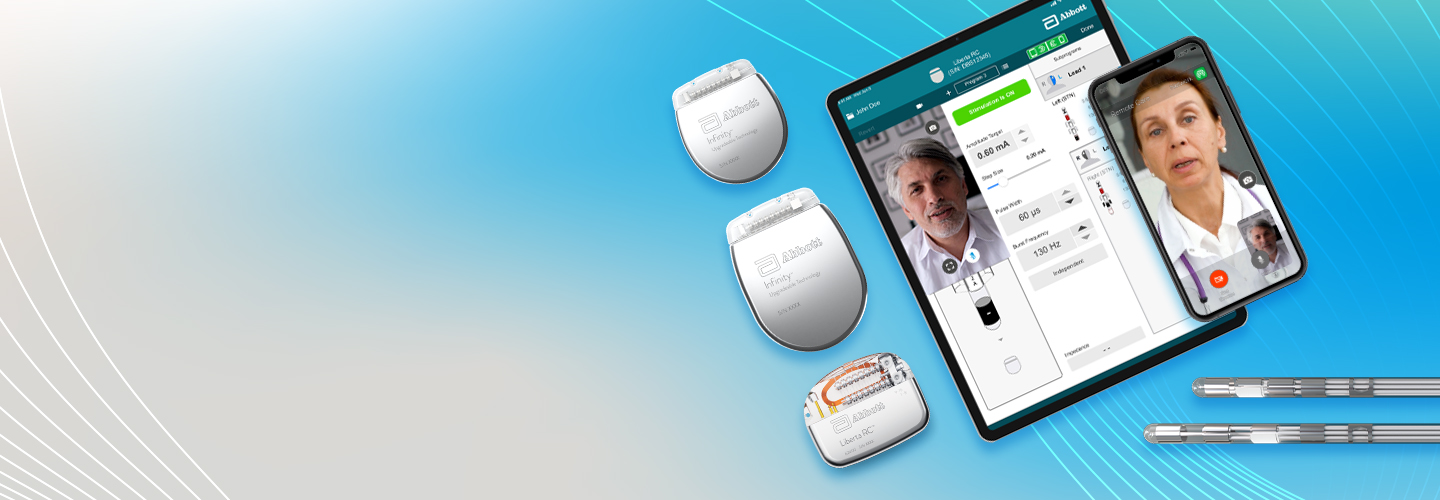
The Liberta RC™ DBS System
With the longest-lasting time between charges and the smallest rechargeable DBS IPG on the market, the Liberta RC™ DBS System looks to set a new standard in convenience and comfort.1,2*,**,***
Built to integrate with our NeuroSphere™ Digital Health platform, the Liberta RC™ DBS System gives you remote access to your doctor, allowing them to adjust your therapy settings, provide real-time charging insights, and more - all from the comfort of your home.†
Benefits:
- Lasts up to 37 days on a full charge - making it the longest time between charges on the market.2,3**
- Smallest FDA-approved rechargeable DBS IPG on the market, offering a discreet implant profile designed for comfort.1*,***
- Only rechargeable DBS device from Abbott in the U.S. that offers remote programming capabilities to healthcare providers for therapy adjustments.1
- Equipped with a wireless charger with enough power for two full recharges.4
The Infinity™ DBS System
With next-generation energy management and an updated longevity profile, the next generation Infinity™ DBS System offers long-lasting, low-maintenance therapy.5
Seamlessly integrated with our NeuroSphere™ Digital Health platform, the Infinity™ DBS System allows you to use your personal Apple‡ iPhone or Abbott‡ provided mobile device and access on-demand support from the comfort of home.†,†††
Benefits:
- Delivers an updated longevity profile thanks to changes in energy management, helping to provide consistent therapy.5††
- Expands patient controller options by letting patients use their personal Apple‡ iPhone or an Abbott‡ provided mobile device.6†††
- Integrates the new NeuroSphere™ Digital Health app, offering on-demand support tools, a comprehensive educational library, and guided tutorial videos to help simplify therapy management.6
Manage your DBS journey with NeuroSphere™ Digital Health
NeuroSphere™ Digital Health app provides access to on-demand education and support resources, live product support, and virtual programming adjustments.
Access on-demand education and support resources early in your journey to understand what to expect ahead.
Interact with our ambassadors and Therapy Navigation Center to get questions answered and gain confidence in your therapy journey.
Access clinician support with remote programming capabilities to help optimize therapy faster than traditional in-clinic appointments.7§

Expecting to have other surgeries in the future?
Surgery Mode helps protect your implanted DBS system from damage during a surgical procedure. For Infinity™ DBS systems, Surgery Mode must be turned on using the Patient Controller prior to surgery. For instructions on how to do this, click here.
If the implant is damaged during surgery, you will need another procedure to replace it.
Connect with a DBS ambassador
Connect with people who have real-life experience and are living with DBS through the Abbott DBS ambassador program.

Art of Parkinson's
View our gallery of original artwork and stories from some people with Parkinson's, sharing how they've reclaimed parts of themselves with DBS therapy.
MAT-2603146 v1.0 | Item approved for U.S. use only.


*Patient comfort resulting from reduced features that could lead to discomfort (i.e. sharp edges) and reduced size compared to on-market IPGs.
**Upon implant of the Liberta RC™ DBS System, 37 days of therapy when programmed with standard (nominal) stimulation settings as described in device instructions for use. Recommended recharge frequency and duration for competitor rechargeable DBS systems described in their respective IFU or clinical studies, which may involve different patient populations and other variables. Not a head-to-head comparison of stimulation settings or clinical outcomes.
***Comparison is limited to the US market only and is based on volume measurements of the following smallest IPG offerings: Abbott Liberta RC™ DBS System (13.6 cc), Boston Scientific‡ Vercise Genus‡ R16 (20.1 cc), and Medtronic‡ Percept RC‡ (13.77 cc). Sizing for the Liberta RC™ DBS IPG is determined using engineering model measurement(s), and methods to calculate size may vary among manufacturers.
†Anywhere with a cellular or Wi-Fi connection and sufficiently charged patient controller and neurostimulation device.
††Individual results may vary. See Clinician’s Manual for battery longevity information.
†††Available on eligible mobile devices. For a list of mobile devices compatible with Abbott's Neuromodulation Patient Controller applications, visit http://www.NMmobiledevicesync.com/dbs
§Anywhere with a WiFi‡ or cellular connection and sufficiently charged battery.
§§With more rapid availability of clinicians for program optimization using NeuroSphere™ Virtual Clinic in the first 90 days after initial programming.
- Abbott. Liberta RCTM DBS System Size Comparison Claims Memo (MAT-2400042). 2025.
- Abbott. Liberta RC™ DBS System Recharging Comparison Memo Claims Memo (MAT-2400043). 2024.
- Abbott. Liberta RC™ Implantable Pulse Generator Clinician's Manual. Plano, TX. 2024.
- Abbott. Jupiter Software Formula Specification. Plano, TX. 2023.
- Abbott. Next Generation Infinity™ DBS System Memo (MAT-2501040). 2025
- Abbott. Hercules Claims Memo (MAT-2501025). 2025
- Gharabaghi A, Groppa S, Navas-Garcia M, et al. Accelerated symptom improvement in Parkinson’s disease via remote internet-based optimization of deep brain stimulation therapy: a randomized controlled multicenter trial. Commun Med (Lond). 2025;5(1):31. doi:10.1038/s43856-025-00744-7. (n = 98 subjects.)
Important Safety Information
Deep Brain Stimulation (DBS) Therapy
Intended Use
This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. The system is intended to be used with leads and associated extensions that are compatible with the system.
Indications For Use
The Abbott Medical deep brain stimulation system is indicated for the following conditions:
- Bilateral stimulation of the subthalamic nucleus (STN) or the internal globus pallidus (GPi) as an adjunctive therapy to reduce some of the symptoms of advanced levodopa-responsive Parkinson’s disease that are not adequately controlled by medications.
- Unilateral or bilateral stimulation of the ventral intermediate nucleus (VIM) of the thalamus for the suppression of disabling upper extremity tremor in adult essential tremor patients whose tremor is not adequately controlled by medications and where the tremor constitutes a significant functional disability.
Contraindications
This system is contraindicated for patients who meet the following criteria:
Are unable to operate the system
Have unsuccessful test stimulation
The following procedures are contraindicated for patients with a deep brain stimulation system. Advise patients to inform their healthcare professional that they cannot undergo the following procedures:
Diathermy (short-wave diathermy, microwave diathermy, or therapeutic ultrasound diathermy)
Electroshock therapy and transcranial magnetic stimulation (TMS)
MRI Safety Information
Some models of this system are Magnetic Resonance (MR) Conditional, and patients with these devices may be scanned safely with magnetic resonance imaging (MRI) when the conditions for safe scanning are met. For more information about MR Conditional neurostimulation components and systems, including equipment settings, scanning procedures, and a complete listing of conditionally approved components, refer to the MRI procedures clinician's manual for neurostimulation systems (available online at medical.abbott/manuals). For more information about MR Conditional products, visit the Abbott Medical product information page at neuromodulation.abbott/MRI-ready.
Warnings
The following warnings apply to this neurostimulation system.
Pregnancy and nursing. Safety and effectiveness of neurostimulation for use during pregnancy and nursing have not been established. Patients should not use this neurostimulation system if they are pregnant or nursing.
Magnetic resonance imaging (MRI). Some patients may be implanted with the components that make up a Magnetic Resonance (MR) Conditional system, which allows them to receive an MRI scan if all the requirements for the implanted components and for scanning are met. A physician can help determine if a patient is eligible to receive an MRI scan by following the requirements provided by Abbott Medical. Physicians should also discuss any risks of MRI with patients.
Patients without an MR Conditional neurostimulation system should not be subjected to MRI because the electromagnetic field generated by an MRI may damage the device electronics and induce voltage through the lead that could jolt or shock the patient.
High stimulation outputs and charge density limits. Avoid excessive stimulation. A risk of brain tissue damage exists with parameter settings using high amplitudes and wide pulse widths.
High amplitudes and wide pulse widths should only be programmed with due consideration of the warnings concerning charge densities. The system can be programmed to use parameter settings outside the range of those used in the clinical studies. If the programming of stimulation parameters exceeds the charge density limit of 30 μC/cm2, a screen will appear warning you that the charge density is too high. Charge density can be reduced by lowering the stimulation amplitude or pulse width. For more information, see the clinician programmer manual.
Higher amplitudes and wider pulse widths may indicate a system problem or a suboptimal lead placement. Stimulation at high outputs may cause unpleasant sensations or motor disturbances or may render the patient incapable of controlling the patient controller. If unpleasant sensations occur, the device should be turned off immediately using the patient magnet.
Diathermy therapy. Do not use short-wave diathermy, microwave diathermy, or therapeutic ultrasound diathermy (all now referred to as diathermy) on patients implanted with a neurostimulation system. Energy from diathermy can be transferred through the implanted system and cause tissue damage at the location of the implanted electrodes, resulting in severe injury or death.
Diathermy is further prohibited because it may also damage the neurostimulation system components. This damage could result in loss of therapy, requiring additional surgery for system implantation and replacement. Injury or damage can occur during diathermy treatment whether the neurostimulation system is turned on or off.
Risk of depression, suicidal ideations, and suicide. New onset or worsening depression, which may be temporary or permanent, is a risk that has been reported with DBS therapy. Suicidal ideation, suicide attempts, and suicide are events that have also been reported. Therefore, physicians should consider the following:
Preoperatively, assess patients for the risks of depression and suicide. This assessment should consider both the risk of depression and suicide as well as the potential clinical benefits of DBS therapy for the condition being treated.
Postoperatively, actively monitor patients for new or worsening symptoms of depression, suicidal thoughts or behaviors, or changes in mood or impulse control.
If a patient experiences new or worsening depression or suicidal ideation, manage these symptoms appropriately.
Educate patients and caregivers about these potential risks prior to implantation, and be sure that they know about the importance of ongoing support and follow-up, including when to contact their health care provider.
Poor surgical risks. Neurostimulation should not be used on patients who are poor surgical risks or patients with multiple illnesses or active general infections.
Explosive or flammable gasses. Do not use the clinician programmer or patient controller in an environment where explosive or flammable gas fumes or vapors are present. The operation of the clinician programmer or patient controller could cause them to ignite, causing severe burns, injury, or death.
Operation of machinery and equipment. Patients should not operate potentially dangerous machinery, power tools, or vehicles or engage in any activity that could be unsafe if their symptoms were to unexpectedly return.
Pediatric use. Safety and effectiveness of neurostimulation for pediatric use have not been established.
Device components. The use of components not approved for use by Abbott Medical with this system may result in damage to the system and increased risk to the patient.
Device modification. This equipment is not serviceable by the customer. To prevent injury or damage to the system, do not modify the equipment. If needed, return the equipment to Abbott Medical for service.
Application modification. To prevent unintended stimulation, do not modify the generator software in any way. Only apply software updates that are published directly by Abbott Medical.
Electrosurgery. To avoid harming the patient or damaging the neurostimulation system, do not use monopolar electrosurgery devices on patients with implanted neurostimulation systems. Before using an electrosurgery device, place the device in Surgery Mode using the patient controller app or clinician programmer app. Confirm the neurostimulation system is functioning correctly after the procedure.
During implant procedures, if electrosurgery devices must be used, take the following actions:
Use bipolar electrosurgery only.
Complete any electrosurgery procedures before connecting the leads or extensions to the neurostimulator.
- Keep the current paths from the electrosurgery device as far from the neurostimulation system as possible.
- Exit Surgery Mode during intraoperative testing and after the procedure is completed. NOTE: During intraoperative testing, Surgery Mode must be turned off for the neurostimulation system to function correctly.
- Confirm that the neurostimulation system is functioning correctly during the implant procedure and before closing the neurostimulator pocket.
After any surgery, check the neurostimulation system for the following:
- Check the neurostimulator to ensure Surgery Mode has been turned off, even if Surgery Mode was not turned on at the beginning or during the procedure.
- Confirm the neurostimulation system is functioning
Radiofrequency or microwave ablation. Careful consideration should be used before using radiofrequency (RF) or microwave ablation in patients who have an implanted neurostimulation system since safety has not been established. Induced electrical currents may cause heating, especially at the lead electrode site, resulting in tissue damage.
Implanted cardiac systems. Physicians need to be aware of the risk and possible interaction between a neurostimulation system and an implanted cardiac system, such as a pacemaker or defibrillator. Electrical pulses from a neurostimulation system may interact with the sensing operation of an implanted cardiac system, causing the cardiac system to respond inappropriately. To minimize or prevent the implanted cardiac system from sensing the output of the neurostimulation system:
Maximize the distance between the implanted systems;
Verify that the neurostimulation system is not interfering with the functions of the implanted cardiac system; and
Avoid programming either device in a unipolar mode (using the device’s can as an anode) or using neurostimulation system settings that interfere with the function of the implantable cardiac system.
Other active implanted devices. The neurostimulation system may interfere with the normal operation of another active implanted device, such as a pacemaker, defibrillator, or another type of neurostimulator. Conversely, the other active implanted device may interfere with the operation of the neurostimulation system.
Interference with other devices. Some of this system’s electronic equipment, such as the programmer and controller, can radiate radiofrequency (RF) energy that may interfere with other electronic devices, including other active implanted devices. Avoid placing equipment components directly over other electronic devices. To correct the effect of interference with other devices, turn off the equipment or increase the distance between the equipment and the device being affected.
Case damage. If the case of the implantable pulse generator (IPG) is pierced or ruptured, severe burns could result from exposure to battery chemicals.
Cremation. The IPG should be explanted before cremation because the IPG could explode. Return the explanted IPG to Abbott Medical.
Generator disposal. Return all explanted components to Abbott Medical for safe disposal. IPGs contain batteries as well as other potentially hazardous materials. Do not crush, puncture, or burn the IPG because explosion or fire may result.
Product materials. Neurostimulation systems have materials that come in contact or may come in contact with tissue. A physician should determine whether or not a patient may have an allergic reaction to these materials before the system is implanted.
Coagulopathies. Physicians should use extreme care with lead implantation in patients with a heightened risk of intracranial hemorrhage. Physicians should also consider underlying factors, such as previous neurological injury or prescribed medications (anticoagulants), that may predispose a patient to the risk of bleeding.
Low frequencies. Stimulation frequencies at less than 30 Hz may cause tremor to be driven (meaning that tremor occurs at the same frequency as the programmed frequency). For this reason, programming at frequencies less than 30 Hz is not recommended.
Return of symptoms and rebound effect. The abrupt cessation of stimulation for any reason will probably cause disease symptoms to return. In some cases, symptoms may return with a greater intensity than what a patient experienced before system implantation (rebound effect). In rare cases, this can create a medical emergency.
Precautions
The following precautions apply to this neurostimulation system.
General Precautions
Surgeon training. Implanting physicians should be experienced in stereotactic and functional neurosurgery.
Clinician training. Clinicians should be familiar with deep brain stimulation therapy and be experienced in the diagnosis and treatment of the indication for which the deep brain stimulation components are being used.
Patient training. Instruct patients to use their neurostimulation system only after an authorized clinician has programmed the generator and has trained the patient on how to safely control stimulation and to charge the system
Patient selection. Select patients appropriately for deep brain stimulation. The patient should be able and willing to use the patient controller and correctly interpret the icons and messages that appear on the screen.
Especially consider the following additional factors when selecting patients:
Level of available support from a caregiver.
Expected effect from cessation of therapy, should disease symptoms return unexpectedly.
Patient's age, as very young or very old patients may have difficulty performing required monitoring of the device.
Patient's mental capacity, as patients with cognitive impairment or those prone to developing dementia would likely have difficulty performing device-related tasks without assistance.
Patient's physical ability, as patients with higher degrees of motor impairment might have difficulty with the physical requirements of monitoring the device.
Patient's visual ability to read the patient controller screen.
Infection. Follow proper infection control procedures. Infections may require that the device be explanted.
Implantation of two systems. If two systems are implanted, ensure that at least 20 cm (8 in.) separates the implanted generators to minimize unintended interaction with other system components.
Implantation of multiple leads. If multiple leads are implanted, leads and extensions should be routed in close proximity. Nonadjacent leads can possibly create a conduit for stray electromagnetic energy that could cause the patient unwanted stimulation.
Implant heating. While charging the generator, patients may perceive an increase in temperature at the generator site. In patients who have areas of increased sensitivity to heat, consider placing the implant where the patient has normal sensation.
Electromagnetic interference (EMI). Some equipment in home, work, medical, and public environments can generate EMI that is strong enough to interfere with the operation of a neurostimulation system or damage system components. Patients should avoid getting too close to these types of EMI sources, which include the following examples:
Commercial electrical equipment (such as arc welders and induction furnaces),
Communication equipment (such as microwave transmitters and high-power amateur transmitters),
High-voltage power lines,
Radiofrequency identification (RFID) devices,
And some medical procedures (such as therapeutic radiation and electromagnetic lithotripsy).
Programmer use. Allow only authorized use of the clinician programmer to avoid any programming changes that may injure a patient.
Security, antitheft, and radiofrequency identification (RFID) devices. Some antitheft devices, such as those used at entrances or exits of department stores, libraries, and other public places, and airport security screening devices may affect stimulation. Additionally, RFID devices, which are often used to read identification badges, as well as some tag deactivation devices, such as those used at payment counters at stores and loan desks at libraries, may also affect stimulation.
Patients should cautiously approach such devices and should request help to bypass them. If they must go through or near a gate or doorway containing this type of device, patients should move quickly and then check their IPG to determine if it is turned on or off.
Unauthorized changes to stimulation parameters. Caution patients to not make unauthorized changes to physician-established stimulation parameters.
Damage to shallow implants. Falling and other traumatic accidents can damage shallowly implanted components such as the leads and extensions.
Keep programmers and controllers dry. The clinician programmer and patient controller are not waterproof. Keep them dry to avoid damage. Advise patients to not use the patient controller when engaging in activities that might cause it to get wet, such as swimming or bathing.
Handle the programmers and controllers with care. The clinician programmer and patient controllers are sensitive electronic devices that can be damaged by rough handling, such as dropping them on the ground.
Battery care. Batteries can explode, leak, or melt if disassembled, shorted (when battery connections contact metal), or exposed to high temperature or fire.
Long-term safety and effectiveness. The long-term safety and effectiveness of this neurostimulation system has not been established beyond 5 years. Safety and effectiveness has not been established for patients with a neurological disease other than Parkinson’s disease or essential tremor, previous surgical ablation procedures, dementia, coagulopathies, or moderate to severe depression; patients under 22 years; implantation in targets other than the STN for Parkinson's disease and the VIM for essential tremor; patients with an active implantable device; patients requiring MRI.
Sterilization And Storage
Single-use, sterile device. The implanted components of this neurostimulation system are intended for a single use only. Sterile components in this kit have been sterilized using ethylene oxide (EtO) gas before shipment and are supplied in sterile packaging to permit direct introduction into the sterile field. Do not resterilize or reimplant an explanted system for any reason.
Storage environment. Store components and their packaging where they will not come in contact with liquids of any kind. Detailed information on storage environment is provided in the appendix of this manual.
Handling And Implantation
Expiration date. An expiration date (or “use-before” date) is printed on the packaging. Do not use the system if the use-before date has expired.
Recharge. Recharge information is printed on the packaging, which instructs the user to fully charge the generator before implantation.
Handle devices with care. The clinician programmer and patient controller are sensitive electronic devices that can be damaged by rough handling, such as dropping them on the ground.
Care and handling of components. Use extreme care when handling system components. Excessive heat, excessive traction, excessive bending, excessive twisting, or the use of sharp instruments may damage and cause failure of the components.
Package or component damage. Do not implant a device if the sterile package or components show signs of damage, if the sterile seal is ruptured, or if contamination is suspected for any reason. Return any suspect components to Abbott Medical for evaluation.
Exposure to body fluids or saline. Prior to connection, exposure of the metal contacts, such as those on the connection end of a lead or extension, to body fluids or saline can lead to corrosion. If such exposure occurs, clean the affected parts with sterile, deionized water or sterile water for irrigation, and dry them completely prior to lead connection and implantation.
Skin erosion. To avoid the risk of skin erosion, implant components at the appropriate depth and inform patients to avoid touching their skin where components are implanted. The IPG should be placed into the pocket, at a depth not to exceed 4.0 cm (1.57 in), with the logo side facing toward the skin surface.
System testing. To ensure correct operation, always test the system during the implant procedure, before closing the neurostimulator pocket, and before the patient leaves the surgery suite.
Multiple leads. When multiple leads are implanted, route the lead extensions so the area between them is minimized. If the lead extensions are routed in a loop, the loop will increase the potential for electromagnetic interference (EMI).
Abandoned leads and replacement leads. The long-term safety associated with multiple implants, leads left in place without use, replacement of leads, multiple implants into the target structure, and lead explant is unknown.
Placement of lead connection in neck. The lead-extension connector should not be placed in the soft tissues of the neck due to an increased incidence of lead fracture.
Hospital And Medical Environments
Electrical medical treatment. In the case that a medical treatment is administered where an electrical current is passed through the body from an external source, first deactivate the IPG by setting all electrodes to off, turning stimulation off, and setting amplitude to zero. Regardless if the device is deactivated, take care to monitor the device for proper function during and after treatment.
High-output ultrasonics and lithotripsy. The use of high-output devices, such as an electrohydraulic lithotriptor, may cause damage to the electronic circuitry of an implanted IPG. If lithotripsy must be used, do not focus the energy near the IPG.
Ultrasonic scanning equipment. The use of ultrasonic scanning equipment may cause mechanical damage to an implanted neurostimulation system if used directly over the implanted system.
External defibrillators. The safety of discharge of an external defibrillator on patients with implanted neurostimulation systems has not been established.
Therapeutic radiation. Therapeutic radiation may damage the electronic circuitry of an implanted neurostimulation system, although no testing has been done and no definite information on radiation effects is available. Sources of therapeutic radiation include therapeutic X rays, cobalt machines, and linear accelerators. If radiation therapy is required, the area over the implanted IPG should be shielded with lead. Damage to the system may not be immediately detectable.
Electrocardiograms. Ensure the neurostimulator is off before initiating an electrocardiogram (ECG). If the neurostimulator is on during an ECG, the ECG recording may be adversely affected, resulting in inaccurate ECG results. Inaccurate ECG results may lead to inappropriate treatment of the patient.
Home And Occupational Environments
Patient activities and environmental precautions. Patients should take reasonable care to avoid devices that generate strong EMI, which may cause the neurostimulation system to unintentionally turn on or off. Patients should also avoid any activities that would be potentially unsafe if their symptoms were to return unexpectedly. These activities include but are not limited to climbing ladders and operating potentially dangerous machinery, power tools, and vehicles. Sudden loss of stimulation may cause patients to fall or lose control of equipment or vehicles, injure others, or bring injury upon themselves.
Control of the patient controller. Advise patients to keep the patient controller away from children and pets in order to avoid potential damage or other hazards.
Activities requiring excessive twisting or stretching. Patients should avoid activities that may put undue stress on the implanted components of the neurostimulation system. Activities that include sudden, excessive or repetitive bending, twisting, or stretching can cause component fracture or dislodgement. Component fracture or dislodgement may result in loss of stimulation, intermittent stimulation, stimulation at the fracture site, and additional surgery to replace or reposition the component.
Activities requiring coordination. Loss of coordination is a potential side effect of DBS therapy. Patients should exercise reasonable caution when participating in activities that require coordination, including those that they were able to perform prior to receiving DBS therapy (for example, swimming).
Bathing. Patients should exercise reasonable caution when bathing.
Component manipulation by patient. Advise your patient to avoid manipulating the implanted system components (e.g., the neurostimulator, the burr hole site). This can result in component damage, lead dislodgement, skin erosion, or stimulation at the implant site. Manipulation may cause device inversion, inhibiting the ability to use the magnet to start or stop stimulation.
Scuba diving or hyperbaric chambers. Patients should not dive below 30 m (100 ft) of water or enter hyperbaric chambers above 4.0 atmospheres absolute (ATA). Pressures below 30 m (100 ft) of water (or above 4.0 ATA) could damage the neurostimulation system. Before diving or using a hyperbaric chamber, patients should discuss the effects of high pressure with their physician.
Skydiving, skiing, or hiking in the mountains. High altitudes should not affect the neurostimulator; however, the patient should consider the movements involved in any planned activity and take precautions to avoid putting undue stress on the implanted system. Patients should be aware that during skydiving, the sudden jerking that occurs when the parachute opens may cause lead dislodgement or fractures, which may require surgery to repair or replace the lead.
Wireless use restrictions. In some environments, the use of wireless functions (e.g., Bluetooth® wireless technology) may be restricted. Such restrictions may apply aboard airplanes, near explosives, or in hazardous locations. If you are unsure of the policy that applies to the use of this device, please ask for authorization to use it before turning it on. (Bluetooth® is a registered trademark of Bluetooth SIG, Inc.)
Consumer goods and electronic devices. Magnetic interference with consumer goods or electronic devices that contain magnets, such as mobile phones and smart watches, may unintentionally cause the neurostimulation system to turn on or turn off or affect communication between the device and generator however, it will not change the prescribed programmed parameters. Patients should be advised to keep their mobile phones and smart watches at least 15 cm (6 in.) away from the generator and avoid placing any smart device in a pocket near the generator. If a patient is concerned about a smart device interacting with their neurostimulation system, consider disabling magnet mode. For more information about setting the magnet mode, refer to the clinician programmer manual or contact Technical Support.
Household appliances. Household appliances that contain magnets (e.g., refrigerators, freezers, inductive cooktops, stereo speakers, mobile telephones, cordless telephones, standard wired telephones, AM/FM radios, and some power tools) may unintentionally cause the neurostimulation system to turn on or turn off.
Therapeutic magnets. Patients should be advised to not use therapeutic magnets. Therapeutic magnets (e.g., magnets used in pillows, mattress pads, back belts, knee braces, wrist bands, and insoles) may unintentionally cause the neurostimulation system to turn on or off.
Adverse Effects
Deep brain stimulation potentially has the following adverse effects:
Possible surgical complications. Surgical complications include, but are not limited to, the following:
Intracranial hemorrhage (which can lead to stroke, paralysis, or death);
Subcutaneous hemorrhage or seroma;
Hematoma;
Cerebrospinal fluid leakage or cerebrospinal fluid abnormality;
Brain contusion;
Infection or inflammation;
Antibiotic anaphylaxis;
Skin disorder;
Edema;
Persistent pain at surgery site or IPG site;
Erosion;
Brachial plexus injury (nerves to chest, shoulder and arm);
Postoperative pain, stress, or discomfort;
Neuropathy (nerve degeneration);
Hemiparesis (muscular weakness or partial paralysis on one side of body);
Ballism or hemiballism (uncontrollable movements on both or only one side of the body);
Confusion—transient, nocturnal or ongoing;
Cognitive impairment, including delirium, dementia, disorientation, psychosis and speech difficulties;
Aphasia;
Deep vein thrombosis;
Complications from anesthesia;
Phlebitis (vein inflammation);
Pulmonary embolism (sudden blood vessel obstruction);
Aborted procedures (air embolism, unable to find target, surgical complication, etc.);
Complications from unusual physiological variations in patients, including foreign body rejection phenomena;
Pneumonia, seizure or convulsions;
Paralysis (loss of motor function, inability to move);
Stroke and death.
Possible deep brain stimulation complications. Deep brain stimulation complications include, but are not limited to, the following:
Device-related complications
Undesirable changes in stimulation related to cellular changes in tissue around the electrodes, changes in the electrode position, loose electrical connections, or lead fracture
Loss of therapeutic benefit as a result of change in electrode positions, loose electrical connections, or lead or extension fracture
Initial jolt or tingling during stimulation; jolting or shocking sensations
Infection
Paresthesia
Lead fracture, migration, or dislodgement
Misplaced lead
Extension malfunction, fracture, or disconnect
Deep brain stimulation system failure or battery failure within the device
Deep brain stimulation system malfunction or dislodgement
Spontaneous turning on or off of the IPG
Allergic or rejection response to implanted materials
Persistent pain, tightness, or redness at the incision sites or general pain
General erosion or local skin erosion over the IPG
Persistent pain, tightness, or discomfort around the implanted parts (e.g., along the extension path in the neck)
Impaired wound healing (e.g., incision site drainage) or abscess formation
Additional neurosurgical procedure to manage one of the above complications or to replace a malfunctioning component
Stimulation-related complications or other complications
Worsening of motor impairment and Parkinson’s disease symptoms including dyskinesia, rigidity, akinesia or bradykinesia, myoclonus, motor fluctuations, abnormal gait or incoordination, ataxia, tremor, and dysphasia
Paresis, asthenia, hemiplegia, or hemiparesis
Dystonia
Sensory disturbance or impairment including neuropathy, neuralgia, sensory deficit, headache, and hearing and visual disturbance
Speech or language impairment including, aphasia, dysphagia, dysarthria, and hypophonia
Cognitive impairment including attention deficit, confusion, disorientation, abnormal thinking, hallucinations, amnesia, delusions, dementia, inability to act or make decisions, psychic akinesia, long term memory impairment, psychiatric disturbances, depression, irritability or fatigue, mania or hypomania, psychosis, aggression, emotional lability, sleep disturbance, anxiety, apathy, drowsiness, alteration of mentation, postural instability and disequilibrium
Restless leg syndrome
Supranuclear gaze palsy
Hypersexuality or increased libido
Decreased therapeutic response
Urinary incontinence or retention
Diarrhea or constipation
Cardiac dysfunction (e.g., hypotension, heart rate changes, or syncope)
Difficulty breathing
Increased salivation
Weight gain or loss
Eye disorder including eye apraxia or blepharospasm
Nausea or vomiting
Sweating
Fever
Hiccups
Cough
Cramps
Worsening existing medical conditions
23-86540 MAT-2215217 v3.0 | Item approved for U.S. use only.
MAT-2602757 v2.0 | Item approved for U.S. use only.


Chronic Pain